Development of ex vivo system for mesenchymal stem cell differentiation and cartilage integration (Vivoload)
Background
Current culture models to investigate cartilage repair therapies are often highly simplified. Even critical in vivo signals such as kinematic load are lacking. This limits the efficacy of in vitro tests, placing a higher burden on in vivo models.
Goal
This project aims to develop a novel ex vivo culture system, which is more representative of the in vivo articulating joint. Media composition, vis-à-vis synovial fluid, will be considered, as will osteochondral plug development, interaction/signaling between cartilage, bone and implant. Finally, complex multiaxial load will also be applied to produce a mechanical environment more associated with the articulating joint.
Results
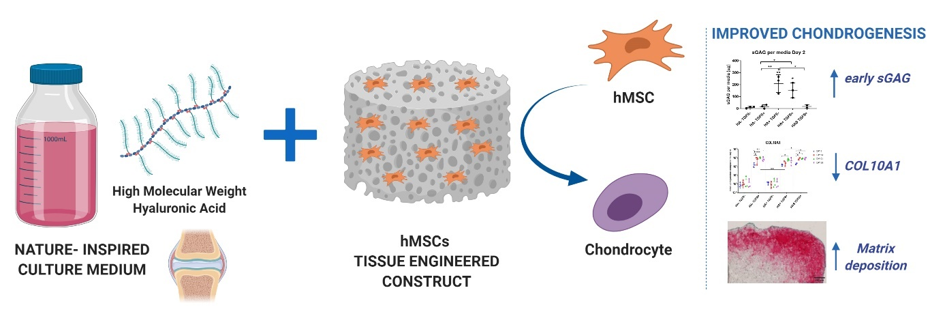
Adding hyaluronic acid, either in the culture medium or mixed within the fibrin scaffold, leads to a more stable chondrogenic phenotype and an increased deposition of cartilage like matrix. We have also shown that the chondrogenic signal is in part generated by the mechanical induction and activation of TGF-β growth factor. This can be used as a new outcome measure to assess novel biomaterials for cartilage regeneration. We expect the incorporation of a more viscous physiological culture medium to modulate the chondrogenic induction of human mesenchymal stem cells induced by interfacial shear. New 3 layer de novo implants have been produced and subjected to complex load, allowing for the development of signaling gradients to be developed under defined conditions. Confining the implant within an osteochondral defect will also modify the response due to paracrine signaling from the viable cartilage and underlying bone. In addition, there is the potential for cell migration from the surrounding "host" tissue, which may also influence the response. Each of the conditions being modified is to bring the in vitro situation nearer to that found in vivo.
-
Publication
Fahy N, Menzel U, Alini M, Stoddart MJ. Shear and Dynamic Compression Modulates the Inflammatory Phenotype of Human Monocytes in vitro. Front Immunol. 2019 Mar 5;10:383. doi: 10.3389/fimmu.2019.00383.
Armiento AR, Alini M, Stoddart MJ. Articular fibrocartilage - Why does hyaline cartilage fail to repair? Adv Drug Deliv Rev. 2019 Jun;146:289-305. doi: 10.1016/j.addr.2018.12.015
Glatt V, Evans CH, Stoddart MJ. Regenerative Rehabilitation: The role of mechanotransduction in orthopaedic regenerative medicine. J Orthop Res. 2019;37(6):1263-9
Monaco G, El Haj AJ, Alini M, Stoddart MJ. Ex vivo systems to study chondrogenic differentiation and cartilage Integration. J Funct Morphol Kinesiol. 2021;6(1):6. https://doi.org/10.3390/jfmk6010006
Monaco G, El Haj AJ, Alini M, Stoddart MJ. Sodium hyaluronate supplemented culture medium combined with joint-simulating mechanical loading improves chondrogenic differentiation of human mesenchymal stem cells. Eur Cell Mater. 2021;41:616-32Monaco G, Qawasmi F, El Haj AJ, Forsyth NR, Stoddart MJ. Chondrogenic differentiation of human bone marrow MSCs in osteochondral implants under kinematic mechanical load is dependent on the underlying osteo component. Front Bioeng Biotechnol. 2022;10:998774. https://doi.org/10.3389/fbioe.2022.998774
-
Presentation
Monaco G, Alini M, El Haj A, Stoddart MJ. Hyaluronan supplemented culture media significantly increases early chondrogenesis glycosaminoglycan synthesis and reduces the upregulation of collagen X in a stem cell-based implant. 2019 TERMIS EU (oral)
Stoddart MJ. Role of kinematic load on cell behavior. 2019 TERMIS EU (oral)
Stoddart MJ. MSCs and their potential for orthopaedic applications. The Joint Seminar Series, Rush University Medical Center, Chicago, USA. (oral)
Stoddart MJ. Role of kinematic load on cell behaviour. TERMISEU2019, Greece. (oral)Stoddart MJ. Physical modulation to divert stem cells to chondrogenic cell fate, ORS2019, Austin, Texas, USA. (oral)
-
Partner
El Haj A (Prof) University of Birmingham, UK